Yane's Site
My classwork from BIMM143
Class 18: Pertussis Mini-Project
Yane (PID A17670350)
Background
Pertussis (whooping cough) is a common lung infection cause by the bacteria B. Pertussis. It can infect everyone but is most deadly for infants (under 1 year of age)
#CDC tracking data The CDC tracks the number of Pertussis cases:
Q. I want a plot of year vs cases
library(ggplot2)
ggplot(cdc) +
aes(year, cases) +
geom_point() +
geom_line() +
geom_vline(xintercept = 1946, col="blue", lty=2) +
geom_vline(xintercept = 1996, col="red", lty=2) +
geom_vline(xintercept = 2020, col="gray", lty=2)
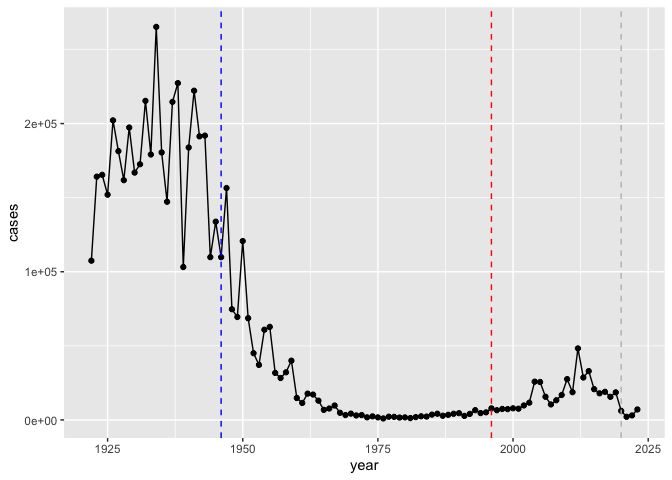
After 1946, the cases of Pertussis decrease as a result of the wP vaccine. In 1996, after the switch to the aP vaccine there was a slight increase in cases. The aP vaccine was created to have less side effects, and this, along with the increase of cases after 1996, suggests that the aP vaccine could be slightly weaker than the wP vaccine.
Q. Add annotation lines for the major milestones of wP vaccination roll-out (1946) and the switch to the aP vaccine (1996).
cdc <- data.frame( year = c(1922L,1923L,1924L,1925L,
1926L,1927L,1928L,1929L,1930L,1931L,
1932L,1933L,1934L,1935L,1936L,
1937L,1938L,1939L,1940L,1941L,1942L,
1943L,1944L,1945L,1946L,1947L,
1948L,1949L,1950L,1951L,1952L,
1953L,1954L,1955L,1956L,1957L,1958L,
1959L,1960L,1961L,1962L,1963L,
1964L,1965L,1966L,1967L,1968L,1969L,
1970L,1971L,1972L,1973L,1974L,
1975L,1976L,1977L,1978L,1979L,1980L,
1981L,1982L,1983L,1984L,1985L,
1986L,1987L,1988L,1989L,1990L,
1991L,1992L,1993L,1994L,1995L,1996L,
1997L,1998L,1999L,2000L,2001L,
2002L,2003L,2004L,2005L,2006L,2007L,
2008L,2009L,2010L,2011L,2012L,
2013L,2014L,2015L,2016L,2017L,2018L,
2019L,2020L,2021L,2022L,2023L, 2024L,
2025L),
cases = c(107473,164191,165418,152003,
202210,181411,161799,197371,
166914,172559,215343,179135,265269,
180518,147237,214652,227319,103188,
183866,222202,191383,191890,109873,
133792,109860,156517,74715,69479,
120718,68687,45030,37129,60886,
62786,31732,28295,32148,40005,
14809,11468,17749,17135,13005,6799,
7717,9718,4810,3285,4249,3036,
3287,1759,2402,1738,1010,2177,2063,
1623,1730,1248,1895,2463,2276,
3589,4195,2823,3450,4157,4570,
2719,4083,6586,4617,5137,7796,6564,
7405,7298,7867,7580,9771,11647,
25827,25616,15632,10454,13278,
16858,27550,18719,48277,28639,32971,
20762,17972,18975,15609,18617,
6124,2116,3044,7063, 22538,
21906)
)
Exploring CMI-PB data
The CMI-PB project’s https://www.cmi-pb.org/ mission is to provide the scientific community with a comprehensive, high-quality and freely accessible resources of Pertussis booster vaccination.
Basically, make available a large dataset of the immune response to Pertussis. They use a “booster” vaccination as a proxy for Pertussis infection.
They make their data available as JSON format API. We can read this into
R with the read_json() function from the jsonlite package:
library(jsonlite)
subject <- read_json("https://www.cmi-pb.org/api/v5_1/subject",
simplifyVector = TRUE)
head(subject)
subject_id infancy_vac biological_sex ethnicity race
1 1 wP Female Not Hispanic or Latino White
2 2 wP Female Not Hispanic or Latino White
3 3 wP Female Unknown White
4 4 wP Male Not Hispanic or Latino Asian
5 5 wP Male Not Hispanic or Latino Asian
6 6 wP Female Not Hispanic or Latino White
year_of_birth date_of_boost dataset
1 1986-01-01 2016-09-12 2020_dataset
2 1968-01-01 2019-01-28 2020_dataset
3 1983-01-01 2016-10-10 2020_dataset
4 1988-01-01 2016-08-29 2020_dataset
5 1991-01-01 2016-08-29 2020_dataset
6 1988-01-01 2016-10-10 2020_dataset
Q. How many aP and wP individuals are there in this
subjecttable?
table(subject$infancy_vac)
aP wP
87 85
Q. How many male/female are there?
table(subject$biological_sex)
Female Male
112 60
Q. What is the break down of
biological_sexandrace?
Q. Is this representative of the US population?
table(subject$race, subject$biological_sex)
Female Male
American Indian/Alaska Native 0 1
Asian 32 12
Black or African American 2 3
More Than One Race 15 4
Native Hawaiian or Other Pacific Islander 1 1
Unknown or Not Reported 14 7
White 48 32
We can read more tables from the CMI-PB database
specimen <- read_json("https://www.cmi-pb.org/api/v5_1/specimen",
simplifyVector = TRUE)
ab_titer <- read_json("https://www.cmi-pb.org/api/v5_1/plasma_ab_titer",
simplifyVector = TRUE)
head(specimen)
specimen_id subject_id actual_day_relative_to_boost
1 1 1 -3
2 2 1 1
3 3 1 3
4 4 1 7
5 5 1 11
6 6 1 32
planned_day_relative_to_boost specimen_type visit
1 0 Blood 1
2 1 Blood 2
3 3 Blood 3
4 7 Blood 4
5 14 Blood 5
6 30 Blood 6
head(ab_titer)
specimen_id isotype is_antigen_specific antigen MFI MFI_normalised
1 1 IgE FALSE Total 1110.21154 2.493425
2 1 IgE FALSE Total 2708.91616 2.493425
3 1 IgG TRUE PT 68.56614 3.736992
4 1 IgG TRUE PRN 332.12718 2.602350
5 1 IgG TRUE FHA 1887.12263 34.050956
6 1 IgE TRUE ACT 0.10000 1.000000
unit lower_limit_of_detection
1 UG/ML 2.096133
2 IU/ML 29.170000
3 IU/ML 0.530000
4 IU/ML 6.205949
5 IU/ML 4.679535
6 IU/ML 2.816431
To make sense of all this data we need to “join” (a.k.a. “merge” or
“link”) all these tables together. Only then will you know that a given
Ab measurement (from the ab_titer table) was collected on a certain
date (from the specimen table) from a certain wP or aP subject (from
the subject table.
We can use dplyr and the *_join() family of functions to do this.
library(dplyr)
Attaching package: 'dplyr'
The following objects are masked from 'package:stats':
filter, lag
The following objects are masked from 'package:base':
intersect, setdiff, setequal, union
meta <- inner_join(subject, specimen)
Joining with `by = join_by(subject_id)`
head(meta)
subject_id infancy_vac biological_sex ethnicity race
1 1 wP Female Not Hispanic or Latino White
2 1 wP Female Not Hispanic or Latino White
3 1 wP Female Not Hispanic or Latino White
4 1 wP Female Not Hispanic or Latino White
5 1 wP Female Not Hispanic or Latino White
6 1 wP Female Not Hispanic or Latino White
year_of_birth date_of_boost dataset specimen_id
1 1986-01-01 2016-09-12 2020_dataset 1
2 1986-01-01 2016-09-12 2020_dataset 2
3 1986-01-01 2016-09-12 2020_dataset 3
4 1986-01-01 2016-09-12 2020_dataset 4
5 1986-01-01 2016-09-12 2020_dataset 5
6 1986-01-01 2016-09-12 2020_dataset 6
actual_day_relative_to_boost planned_day_relative_to_boost specimen_type
1 -3 0 Blood
2 1 1 Blood
3 3 3 Blood
4 7 7 Blood
5 11 14 Blood
6 32 30 Blood
visit
1 1
2 2
3 3
4 4
5 5
6 6
Let’s do one more inner_join() to join the ab_titer with all our
meta data
abdata <- inner_join(ab_titer, meta)
Joining with `by = join_by(specimen_id)`
head(abdata)
specimen_id isotype is_antigen_specific antigen MFI MFI_normalised
1 1 IgE FALSE Total 1110.21154 2.493425
2 1 IgE FALSE Total 2708.91616 2.493425
3 1 IgG TRUE PT 68.56614 3.736992
4 1 IgG TRUE PRN 332.12718 2.602350
5 1 IgG TRUE FHA 1887.12263 34.050956
6 1 IgE TRUE ACT 0.10000 1.000000
unit lower_limit_of_detection subject_id infancy_vac biological_sex
1 UG/ML 2.096133 1 wP Female
2 IU/ML 29.170000 1 wP Female
3 IU/ML 0.530000 1 wP Female
4 IU/ML 6.205949 1 wP Female
5 IU/ML 4.679535 1 wP Female
6 IU/ML 2.816431 1 wP Female
ethnicity race year_of_birth date_of_boost dataset
1 Not Hispanic or Latino White 1986-01-01 2016-09-12 2020_dataset
2 Not Hispanic or Latino White 1986-01-01 2016-09-12 2020_dataset
3 Not Hispanic or Latino White 1986-01-01 2016-09-12 2020_dataset
4 Not Hispanic or Latino White 1986-01-01 2016-09-12 2020_dataset
5 Not Hispanic or Latino White 1986-01-01 2016-09-12 2020_dataset
6 Not Hispanic or Latino White 1986-01-01 2016-09-12 2020_dataset
actual_day_relative_to_boost planned_day_relative_to_boost specimen_type
1 -3 0 Blood
2 -3 0 Blood
3 -3 0 Blood
4 -3 0 Blood
5 -3 0 Blood
6 -3 0 Blood
visit
1 1
2 1
3 1
4 1
5 1
6 1
Q. How many different Ab “isotype” values are in this dataset?
table(abdata$isotype)
IgE IgG IgG1 IgG2 IgG3 IgG4
6698 7265 11993 12000 12000 12000
Q. How many different “antigen” values are measured?
table(abdata$antigen)
ACT BETV1 DT FELD1 FHA FIM2/3 LOLP1 LOS Measles OVA
1970 1970 6318 1970 6712 6318 1970 1970 1970 6318
PD1 PRN PT PTM Total TT
1970 6712 6712 1970 788 6318
Let’s focus on IgG isotype
igg <- abdata |>
filter (isotype=="IgG")
head(igg)
specimen_id isotype is_antigen_specific antigen MFI MFI_normalised
1 1 IgG TRUE PT 68.56614 3.736992
2 1 IgG TRUE PRN 332.12718 2.602350
3 1 IgG TRUE FHA 1887.12263 34.050956
4 19 IgG TRUE PT 20.11607 1.096366
5 19 IgG TRUE PRN 976.67419 7.652635
6 19 IgG TRUE FHA 60.76626 1.096457
unit lower_limit_of_detection subject_id infancy_vac biological_sex
1 IU/ML 0.530000 1 wP Female
2 IU/ML 6.205949 1 wP Female
3 IU/ML 4.679535 1 wP Female
4 IU/ML 0.530000 3 wP Female
5 IU/ML 6.205949 3 wP Female
6 IU/ML 4.679535 3 wP Female
ethnicity race year_of_birth date_of_boost dataset
1 Not Hispanic or Latino White 1986-01-01 2016-09-12 2020_dataset
2 Not Hispanic or Latino White 1986-01-01 2016-09-12 2020_dataset
3 Not Hispanic or Latino White 1986-01-01 2016-09-12 2020_dataset
4 Unknown White 1983-01-01 2016-10-10 2020_dataset
5 Unknown White 1983-01-01 2016-10-10 2020_dataset
6 Unknown White 1983-01-01 2016-10-10 2020_dataset
actual_day_relative_to_boost planned_day_relative_to_boost specimen_type
1 -3 0 Blood
2 -3 0 Blood
3 -3 0 Blood
4 -3 0 Blood
5 -3 0 Blood
6 -3 0 Blood
visit
1 1
2 1
3 1
4 1
5 1
6 1
Make a plot of MFI_normalised values for all antigen values.
ggplot(igg) +
aes(MFI_normalised, antigen) +
geom_boxplot()
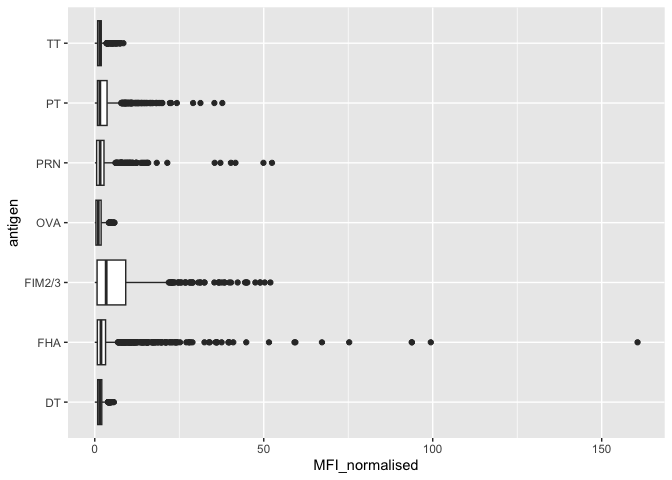
The antigens “PT”, “FIM2/3” and “FHA” appear to have the widest range of values.
Q. Is there a difference for these responses between aP and wP individuals?
ggplot(igg) +
aes(MFI_normalised, antigen, col=infancy_vac) +
geom_boxplot()
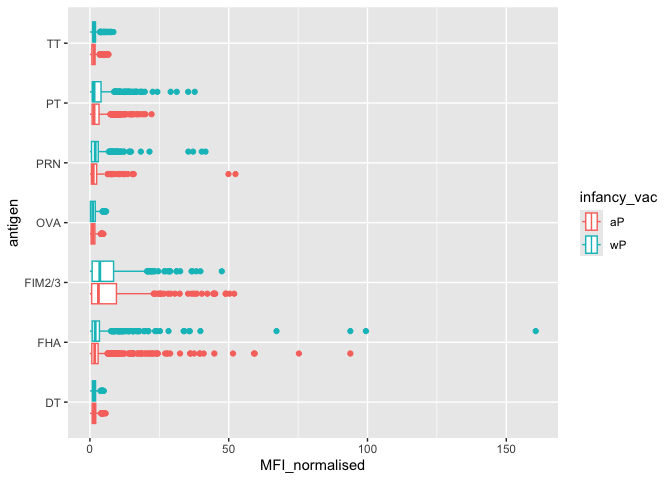
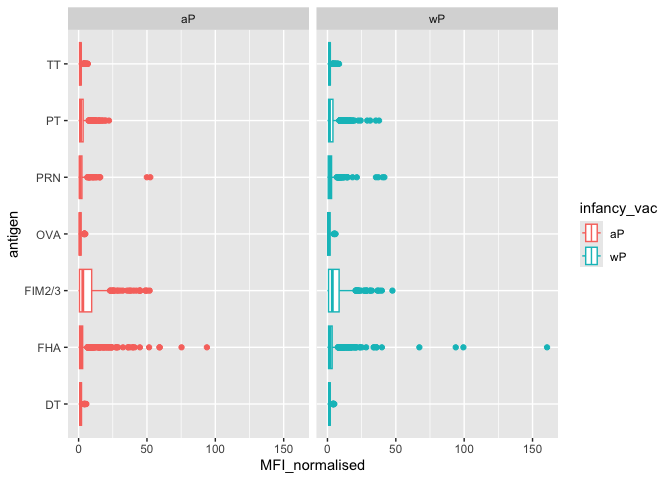
ggplot(igg) +
aes(MFI_normalised, antigen, col=infancy_vac) +
geom_boxplot() +
facet_wrap(~infancy_vac)
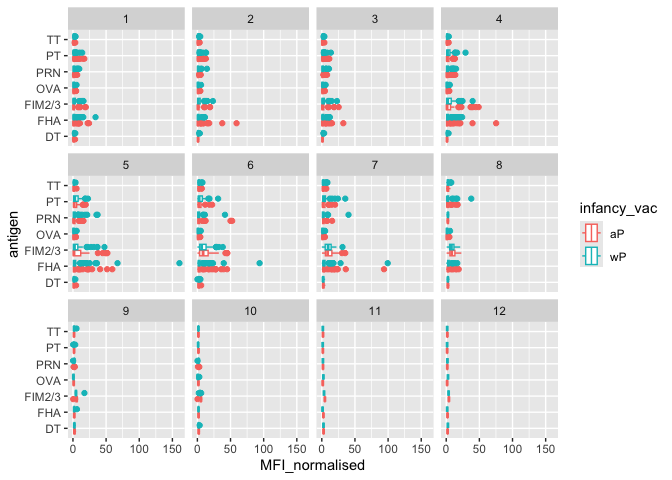
Q. Is there a difference with time (i.e. before booster shot vs after booster shot)
ggplot(igg) +
aes(MFI_normalised, antigen, col=infancy_vac) +
geom_boxplot() +
facet_wrap(~visit)
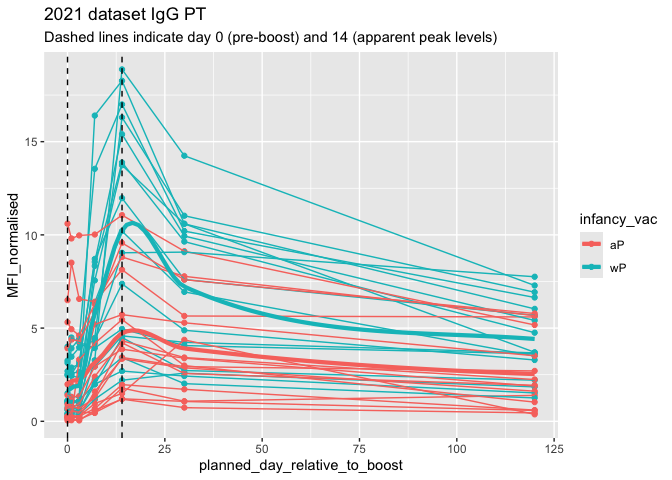
## filter to 2021 dataset, IgG and PT only
abdata.PT.21 <- abdata |>
filter(dataset == "2021_dataset",
isotype == "IgG",
antigen =="PT")
ggplot(abdata.PT.21) +
aes(x=planned_day_relative_to_boost,
y=MFI_normalised,
col=infancy_vac,
group=subject_id) +
geom_point() +
geom_line() +
geom_smooth(aes(group=infancy_vac), se = FALSE, method = "loess",
span = 0.4, linewidth = 1.5) +
geom_vline(xintercept=0, linetype="dashed") +
geom_vline(xintercept=14, linetype="dashed") +
labs(title="2021 dataset IgG PT",
subtitle = "Dashed lines indicate day 0 (pre-boost) and 14 (apparent peak levels)")
`geom_smooth()` using formula = 'y ~ x'
Warning in simpleLoess(y, x, w, span, degree = degree, parametric = parametric,
: pseudoinverse used at -0.6
Warning in simpleLoess(y, x, w, span, degree = degree, parametric = parametric,
: neighborhood radius 3.6
Warning in simpleLoess(y, x, w, span, degree = degree, parametric = parametric,
: reciprocal condition number 0
Warning in simpleLoess(y, x, w, span, degree = degree, parametric = parametric,
: There are other near singularities as well. 11364
Warning in simpleLoess(y, x, w, span, degree = degree, parametric = parametric,
: pseudoinverse used at -0.6
Warning in simpleLoess(y, x, w, span, degree = degree, parametric = parametric,
: neighborhood radius 3.6
Warning in simpleLoess(y, x, w, span, degree = degree, parametric = parametric,
: reciprocal condition number 0
Warning in simpleLoess(y, x, w, span, degree = degree, parametric = parametric,
: There are other near singularities as well. 11364